 
Some theoretical speculations as to what they might be are present in the literature more information can be found at the main articles of the elements involved. As such, unambiguous characterisation of the species they form is impossible, and so their species have been excluded from the table below.  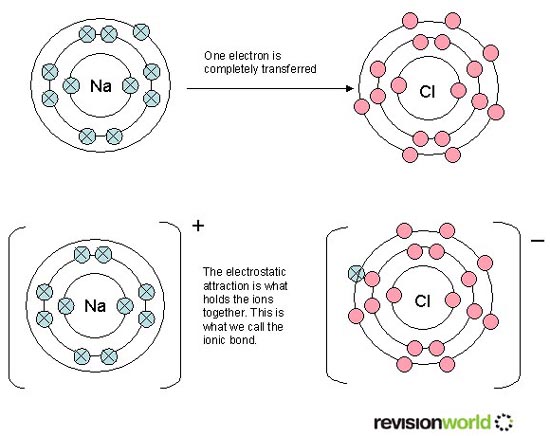
M X n → M n + ( a q ) + n X − ( a q ) ] − List įor some highly radioactive elements, such as astatine and radon, only tracer quantities have been experimented on. ETUDE EXPERIMENTALE D'UN SPECTROMETRE DE MASSE MAGNET I QUE PERMETTANT D ' 480 71 030 NEGATIVE POSITIVE AND NEGATIVE ION MASS SPECTRA OF SOME. Many common materials contain these ions. When a salt of a metal ion, with the generic formula MX n, is dissolved in water, it will dissociate into a cation and anions. Examples of Positive Ions Positive ions are typically metals or act like metals. The existence must be inferred on the basis of indirect evidence provided by modelling with experimental data or by analogy with structures obtained by X-ray crystallography. This table lists the ionic species that are most likely to be present, depending on pH, in aqueous solutions of binary salts of metal ions. the purple manganese oxyanion MnO − 4 from KMnO 4 ( § Mn) Thus, metals are known to have lower electron affinities.the blue copper cation Cu 2+ from CuSO 4 ( § Cu).the turquoise nickel cation Ni 2+ from NiCl 2 ( § Ni).the yellow chromium oxyanion CrO 2− 4 from K 2CrO 4 ( § Cr).the orange chromium oxyanion Cr 2O 2− 7 from K 2Cr 2O 7 ( § Cr) With the exception of hydrogen, all elements that form positive ions by losing electrons during chemical reactions are called metals.the red cobalt cation Co 2+ from Co(NO 3) 2 (see § Co).Metallic ions in aqueous solution display many colours: JSTOR ( November 2021) ( Learn how and when to remove this template message).When a salt such as NaCl dissolves in water, it produces Na + ( aq) and Cl ( aq) ions. Depending on the acidbase properties of its component ions, however, a salt can dissolve in water to produce a neutral solution, a basic solution, or an acidic solution. For binary ionic compounds (ionic compounds that contain only two types of elements), the compounds are named by writing the name of the cation first followed by the name of the anion. Negatively-charged ions are anions, formed from nonmetallic elements like oxygen and sulfur. CH 3CO 2H(l)acid + NaOH(s)baseH 2O H 2OCH 3CO 2Na(aq)salt + H 2O(l)water. Ionic compounds are neutral compounds made up of positively charged ions called cations and negatively charged ions called anions. 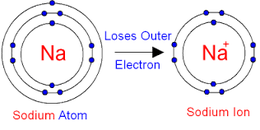
Positive ions are cations and are typically metals like copper or sodium. Unsourced material may be challenged and removed.įind sources: "List of aqueous ions by element" – news Ions electrically charged atoms can carry a positive or negative charge. When these two types of ions meet it results in the destruction of atoms in the process of electrons leaving a positively charged atom and becoming a negatively charged atom. Please help improve this article by adding citations to reliable sources. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding. Electrons are a form of positive ions and protons are a form of negative ion. At this point in your study of chemistry, you should memorize the names, formulas, and charges of the most common polyatomic ions.This article needs additional citations for verification. Oxyanions are polyatomic ions that contain one or more oxygen atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
